Rabbit Anti-Insulin Receptor alpha antibody
CD 220; CD220; CD220 antigen; HHF 5; HHF5; INSR; IR; 4932439J01Rik; ALPHA SUBUNIT INSULIN RECEPTOR; D630014A15Rik; INSULIN RECEPTOR; INSULIN RECEPTOR KINASE; INSULIN RPTK; IR ALPHA; IRK; INSR_HUMAN.
View History [Clear]
Details
Product Name Insulin Receptor alpha Chinese Name 胰岛素受体α抗体 Alias CD 220; CD220; CD220 antigen; HHF 5; HHF5; INSR; IR; 4932439J01Rik; ALPHA SUBUNIT INSULIN RECEPTOR; D630014A15Rik; INSULIN RECEPTOR; INSULIN RECEPTOR KINASE; INSULIN RPTK; IR ALPHA; IRK; INSR_HUMAN. literatures Research Area Cardiovascular Cell biology Neurobiology Signal transduction Growth factors and hormones Kinases and Phosphatases Diabetes The new supersedes the old Immunogen Species Rabbit Clonality Polyclonal React Species Human, Rat, (predicted: Mouse, Chicken, Dog, Pig, Cow, Horse, Rabbit, Sheep, ) Applications WB=1:500-2000 ELISA=1:5000-10000 IHC-P=1:100-500 IHC-F=1:100-500 Flow-Cyt=0.2μg/Test IF=1:100-500 (Paraffin sections need antigen repair)
not yet tested in other applications.
optimal dilutions/concentrations should be determined by the end user.Theoretical molecular weight 80/152kDa Cellular localization The cell membrane Form Liquid Concentration 1mg/ml immunogen KLH conjugated synthetic peptide derived from human Insulin Receptor alpha: 701-760/1382 Lsotype IgG Purification affinity purified by Protein A Buffer Solution 0.01M TBS(pH7.4) with 1% BSA, 0.03% Proclin300 and 50% Glycerol. Storage Shipped at 4℃. Store at -20 °C for one year. Avoid repeated freeze/thaw cycles. Attention This product as supplied is intended for research use only, not for use in human, therapeutic or diagnostic applications. PubMed PubMed Product Detail The human insulin receptor is a heterotetrameric membrane glycoprotein consisting of disulfide linked subunits in a beta-alpha-alpha-beta configuration. The beta subunit (95 kDa) possesses a single transmembrane domain, whereas the alpha subunit (135 kDa) is completely extracellular. The insulin receptor exhibits receptor tyrosine kinase (RTK) activity. RTKs are single pass transmembrane receptors that possess intrinsic cytoplasmic enzymatic activity, catalyzing the transfer of the gamma phosphate of ATP to tyrosine residues in protein substrates. RTKs are essential components of signal transduction pathways that affect cell proliferation, differentiation, migration and metabolism.
Included in this large protein family are the insulin receptor and the receptors for growth factors such as epidermal growth factor, fibroblast growth factor and vascular endothelial growth factor. Receptor activation occurs through ligand binding, which facilitates receptor dimerization and autophosphorylation of specific tyrosine residues in the cytoplasmic portion. The interaction of insulin with the alpha subunit of the insulin receptor activates the protein tyrosine kinase of the beta subunit, which then undergoes an autophosphorylation that increases its tyrosine kinase activity. Three adapter proteins, IRS1, IRS2 and Shc, become phosphorylated on tyrosine residues following insulin receptor activation. These three phosphorylated proteins then interact with SH2 domain containing signaling proteins.
Function:
Receptor tyrosine kinase which mediates the pleiotropic actions of insulin. Binding of insulin leads to phosphorylation of several intracellular substrates, including, insulin receptor substrates (IRS1, 2, 3, 4), SHC, GAB1, CBL and other signaling intermediates. Each of these phosphorylated proteins serve as docking proteins for other signaling proteins that contain Src-homology-2 domains (SH2 domain) that specifically recognize different phosphotyrosines residues, including the p85 regulatory subunit of PI3K and SHP2. Phosphorylation of IRSs proteins lead to the activation of two main signaling pathways: the PI3K-AKT/PKB pathway, which is responsible for most of the metabolic actions of insulin, and the Ras-MAPK pathway, which regulates expression of some genes and cooperates with the PI3K pathway to control cell growth and differentiation. Binding of the SH2 domains of PI3K to phosphotyrosines on IRS1 leads to the activation of PI3K and the generation of phosphatidylinositol-(3, 4, 5)-triphosphate (PIP3), a lipid second messenger, which activates several PIP3-dependent serine/threonine kinases, such as PDPK1 and subsequently AKT/PKB. The net effect of this pathway is to produce a translocation of the glucose transporter SLC2A4/GLUT4 from cytoplasmic vesicles to the cell membrane to facilitate glucose transport. Moreover, upon insulin stimulation, activated AKT/PKB is responsible for: anti-apoptotic effect of insulin by inducing phosphorylation of BAD; regulates the expression of gluconeogenic and lipogenic enzymes by controlling the activity of the winged helix or forkhead (FOX) class of transcription factors. Another pathway regulated by PI3K-AKT/PKB activation is mTORC1 signaling pathway which regulates cell growth and metabolism and integrates signals from insulin. AKT mediates insulin-stimulated protein synthesis by phosphorylating TSC2 thereby activating mTORC1 pathway. The Ras/RAF/MAP2K/MAPK pathway is mainly involved in mediating cell growth, survival and cellular differentiation of insulin. Phosphorylated IRS1 recruits GRB2/SOS complex, which triggers the activation of the Ras/RAF/MAP2K/MAPK pathway. In addition to binding insulin, the insulin receptor can bind insulin-like growth factors (IGFI and IGFII). Isoform Short has a higher affinity for IGFII binding. When present in a hybrid receptor with IGF1R, binds IGF1. PubMed:12138094 shows that hybrid receptors composed of IGF1R and INSR isoform Long are activated with a high affinity by IGF1, with low affinity by IGF2 and not significantly activated by insulin, and that hybrid receptors composed of IGF1R and INSR isoform Short are activated by IGF1, IGF2 and insulin. In contrast, PubMed:16831875 shows that hybrid receptors composed of IGF1R and INSR isoform Long and hybrid receptors composed of IGF1R and INSR isoform Short have similar binding characteristics, both bind IGF1 and have a low affinity for insulin.
Subunit:
Tetramer of 2 alpha and 2 beta chains linked by disulfide bonds. The alpha chains contribute to the formation of the ligand-binding domain, while the beta chains carry the kinase domain. Forms a hybrid receptor with IGF1R, the hybrid is a tetramer consisting of 1 alpha chain and 1 beta chain of INSR and 1 alpha chain and 1 beta chain of IGF1R. Interacts with SORBS1 but dissociates from it following insulin stimulation. Binds SH2B2. Activated form of INSR interacts (via Tyr-999) with the PTB/PID domains of IRS1 and SHC1. The sequences surrounding the phosphorylated NPXY motif contribute differentially to either IRS1 or SHC1 recognition. Interacts (via tyrosines in the C-terminus) with IRS2 (via PTB domain and 591-786 AA); the 591-786 would be the primary anchor of IRS2 to INSR while the PTB domain would have a stabilizing action on the interaction with INSR. Interacts with the SH2 domains of the 85 kDa regulatory subunit of PI3K (PIK3R1) in vitro, when autophosphorylated on tyrosine residues. Interacts with SOCS7. Interacts (via the phosphorylated Tyr-999), with SOCS3. Interacts (via the phosphorylated Tyr-1185, Tyr-1189, Tyr-1190) with SOCS1. Interacts with CAV2 (tyrosine-phosphorylated form); the interaction is increased with 'Tyr-27'phosphorylation of CAV2 (By similarity). Interacts with ARRB2 (By similarity). Interacts with GRB10; this interaction blocks the association between IRS1/IRS2 and INSR, significantly reduces insulin-stimulated tyrosine phosphorylation of IRS1 and IRS2 and thus decreases insulin signaling. Interacts with GRB7 (By similarity). Interacts with PDPK1. Interacts (via Tyr-1190) with GRB14 (via BPS domain); this interaction protects the tyrosines in the activation loop from dephosphorylation, but promotes dephosphorylation of Tyr-999, this results in decreased interaction with, and phosphorylation of, IRS1. Interacts (via subunit alpha) with ENPP1 (via 485-599 AA); this interaction blocks autophosphorylation. Interacts with PTPRE; this interaction is dependent of Tyr-1185, Tyr-1189 and Tyr-1190 of the INSR. Interacts with STAT5B (via SH2 domain). Interacts with PTPRF.
Subcellular Location:
Membrane; Single-pass type I membrane protein.
Tissue Specificity:
Isoform Long and isoform Short are predominantly expressed in tissue targets of insulin metabolic effects: liver, adipose tissue and skeletal muscle but are also expressed in the peripheral nerve, kidney, pulmonary alveoli, pancreatic acini, placenta vascular endothelium, fibroblasts, monocytes, granulocytes, erythrocytes and skin. Isoform Short is preferentially expressed in fetal cells such as fetal fibroblasts, muscle, liver and kidney. Found as a hybrid receptor with IGF1R in muscle, heart, kidney, adipose tissue, skeletal muscle, hepatoma, fibroblasts, spleen and placenta (at protein level). Overexpressed in several tumors, including breast, colon, lung, ovary, and thyroid carcinomas.
Post-translational modifications:
After being transported from the endoplasmic reticulum to the Golgi apparatus, the single glycosylated precursor is further glycosylated and then cleaved, followed by its transport to the plasma membrane.
Autophosphorylated on tyrosine residues in response to insulin. Phosphorylation of Tyr-999 is required for IRS1-, SHC1-, and STAT5B-binding. Dephosphorylated by PTPRE on Tyr-999, Tyr-1185, Tyr-1189 and Tyr-1190 residues. Dephosphorylated by PTPRF.
DISEASE:
Defects in INSR are the cause of Rabson-Mendenhall syndrome (RMS) [MIM:262190]; also known as Mendenhall syndrome. RMS is a severe insulin resistance syndrome characterized by insulin-resistant diabetes mellitus with pineal hyperplasia and somatic abnormalities. Typical features include coarse, senile-appearing facies, dental and skin abnormalities, abdominal distension, and phallic enlargement. Inheritance is autosomal recessive.
Defects in INSR are the cause of leprechaunism (LEPRCH) [MIM:246200]; also known as Donohue syndrome. Leprechaunism represents the most severe form of insulin resistance syndrome, characterized by intrauterine and postnatal growth retardation and death in early infancy. Inheritance is autosomal recessive.
Defects in INSR may be associated with noninsulin-dependent diabetes mellitus (NIDDM) [MIM:125853]; also known as diabetes mellitus type 2.
Defects in INSR are the cause of familial hyperinsulinemic hypoglycemia type 5 (HHF5) [MIM:609968]. Familial hyperinsulinemic hypoglycemia [MIM:256450], also referred to as congenital hyperinsulinism, nesidioblastosis, or persistent hyperinsulinemic hypoglycemia of infancy (PPHI), is the most common cause of persistent hypoglycemia in infancy and is due to defective negative feedback regulation of insulin secretion by low glucose levels.
Defects in INSR are the cause of insulin-resistant diabetes mellitus with acanthosis nigricans type A (IRAN type A) [MIM:610549]. This syndrome is characterized by the association of severe insulin resistance (manifested by marked hyperinsulinemia and a failure to respond to exogenous insulin) with the skin lesion acanthosis nigricans and ovarian hyperandrogenism in adolescent female subjects. Women frequently present with hirsutism, acne, amenorrhea or oligomenorrhea, and virilization. This syndrome is different from the type B that has been demonstrated to be secondary to the presence of circulating autoantibodies against the insulin receptor.
Similarity:
Belongs to the protein kinase superfamily. Tyr protein kinase family. Insulin receptor subfamily.
Contains 3 fibronectin type-III domains.
Contains 1 protein kinase domain.
SWISS:
P06213
Gene ID:
3643
Database links:Entrez Gene: 3643 Human
Entrez Gene: 16337 Mouse
Omim: 147670 Human
SwissProt: P06213 Human
SwissProt: P15208 Mouse
Unigene: 465744 Human
Unigene: 9876 Rat
Product Picture
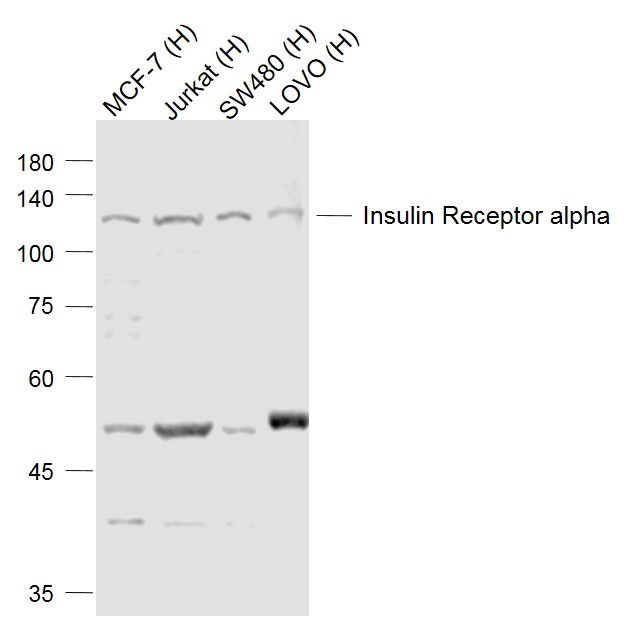
Lane 1: MCF-7 (Human) Cell Lysate at 30 ug
Lane 2: Jurkat (Human) Cell Lysate at 30 ug
Lane 3: SW480 (Human) Cell Lysate at 30 ug
Lane 4: LOVO (Human) Cell Lysate at 30 ug
Primary: Anti-Insulin Receptor alpha (SL0047R) at 1/500 dilution
Secondary: IRDye800CW Goat Anti-Rabbit IgG at 1/20000 dilution
Predicted band size: 120 kD
Observed band size: 120 kD
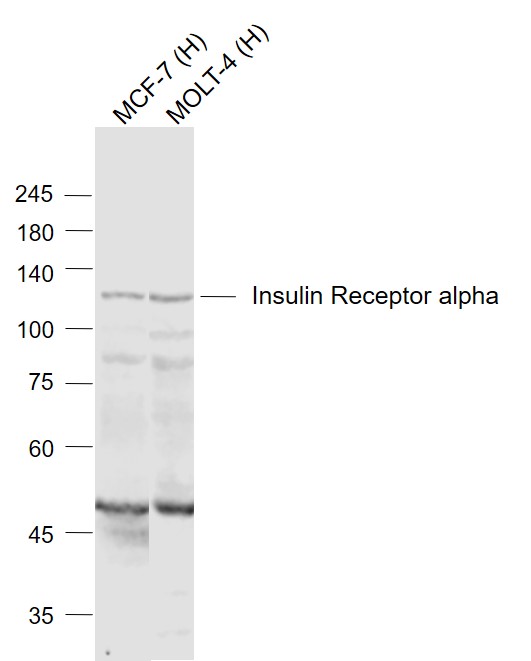
Sample:
Lane 1: MCF-7 (Human) Cell Lysate at 30 ug
Lane 2: MOLT-4 (Human) Cell Lysate at 30 ug
Primary: Anti-Insulin Receptor alpha (SL0047R) at 1/1000 dilution
Secondary: IRDye800CW Goat Anti-Rabbit IgG at 1/20000 dilution
Predicted band size: 120 kD
Observed band size: 120 kD
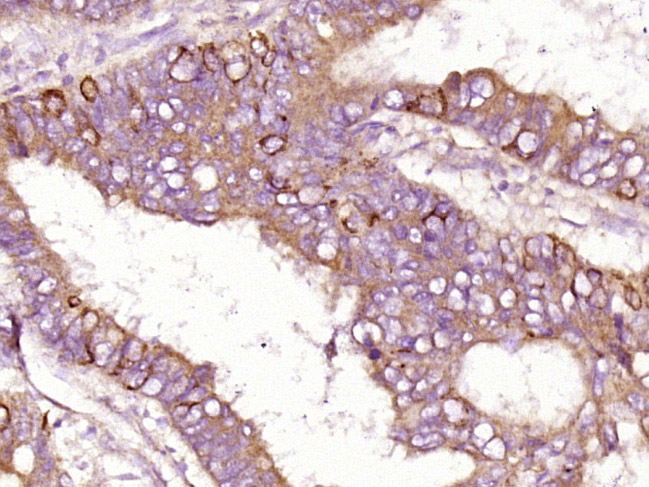
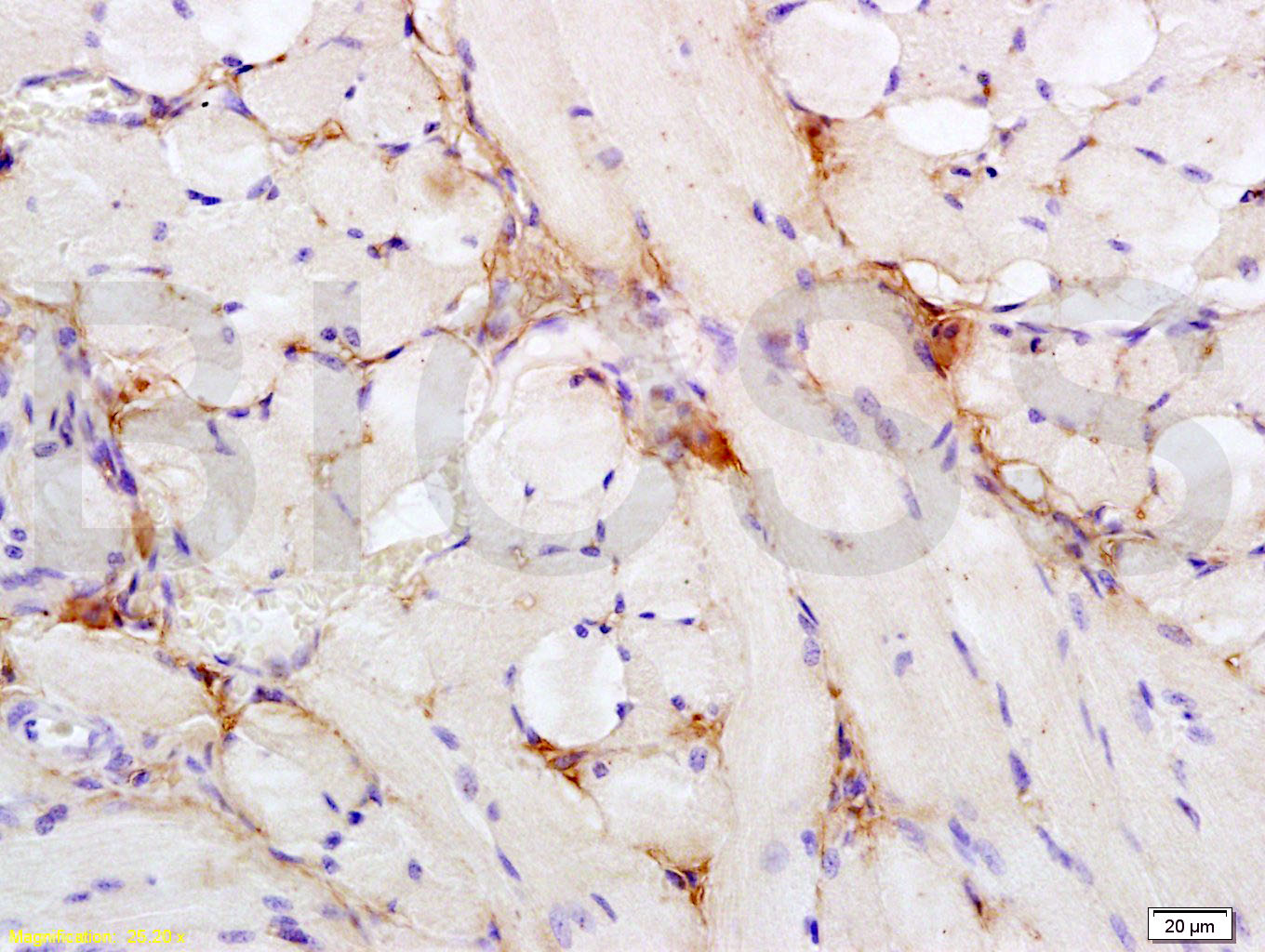
Paraformaldehyde-fixed, paraffin embedded (human colon carcinoma); Antigen retrieval by boiling in sodium citrate buffer (pH6.0) for 15min; Block endogenous peroxidase by 3% hydrogen peroxide for 20 minutes; Blocking buffer (normal goat serum) at 37°C for 30min; Antibody incubation with (Insulin Receptor alpha) Polyclonal Antibody, Unconjugated (SL0047R) at 1:400 overnight at 4°C, followed by operating according to SP Kit(Rabbit) (sp-0023) instructionsand DAB staining.Tissue/cell: rat tongue tissue; 4% Paraformaldehyde-fixed and paraffin-embedded;
Antigen retrieval: citrate buffer ( 0.01M, pH 6.0 ), Boiling bathing for 15min; Block endogenous peroxidase by 3% Hydrogen peroxide for 30min; Blocking buffer (normal goat serum,C-0005) at 37℃ for 20 min;
Incubation: Anti- Insulin Receptor alpha Polyclonal Antibody, Unconjugated(SL0047R) 1:200, overnight at 4°C, followed by conjugation to the secondary antibody(SP-0023) and DAB(C-0010) staining
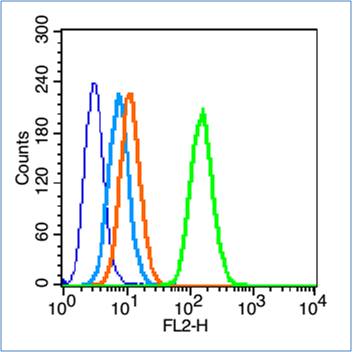
Blank control (blue line): HL60(blue).
Primary Antibody (green line): Rabbit Anti-Insulin Receptor alpha antibody (SL0047R)
Dilution: 0.2μg /10^6 cells;
Isotype Control Antibody (orange line): Rabbit IgG .
Secondary Antibody (white blue line): Goat anti-rabbit IgG-PE
Dilution: 1μg /test.
Protocol
The cells were fixed with 70% ethanol Overnight at 4℃. Cells stained with Primary Antibody for 30 min at room temperature. The cells were then incubated in 1 X PBS/2%BSA/10% goat serum to block non-specific protein-protein interactions followed by the antibody for 15 min at room temperature. The secondary antibody used for 40 min at room temperature. Acquisition of 20,000 events was performed.
Partial purchase records(bought amounts latest0)
No one bought this product
User Comment(Total0User Comment Num)
- No comment


 +86 571 56623320
+86 571 56623320




